Nanoparticle-mediated photothermal therapy uses special nanoparticles, like gold or carbon-based ones, to absorb light and convert it into heat, targeting and destroying cancer cells precisely. This approach offers advantages over traditional treatments, including better targeting and fewer side effects. While it shows great promise, challenges like safety and manufacturing need addressing. To learn how researchers are overcoming these hurdles and advancing this innovative cancer treatment, keep exploring further.
Key Takeaways
- Nanoparticles such as gold nanostructures and carbon-based materials absorb light and convert it into heat for targeted cancer treatment.
- Tailored nanoparticle properties enhance light absorption efficiency and improve therapeutic outcomes in photothermal therapy.
- Surface modifications enable active targeting of tumor cells, increasing treatment precision and minimizing side effects.
- Challenges include ensuring nanoparticle biocompatibility, controlled delivery, and overcoming regulatory hurdles for clinical use.
- Advances focus on designing safer, more effective nanoparticles with improved targeting, stability, and safety profiles for clinical applications.
gold nanoparticle photothermal therapy kit
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
Types of Nanoparticles Used in PTT

When it comes to photothermal therapy (PTT), the choice of nanoparticles is crucial for effective treatment. You need to consider how nanoparticles are synthesized, as different methods influence their size, shape, and stability—factors critical for performance. Common types include gold nanostructures, carbon-based nanoparticles, and metal sulfides, each offering unique properties. Equally important are biocompatibility assessments; ensuring that these nanoparticles are safe for use in the body is essential. These evaluations help determine potential toxicity and immune responses, guiding safer design choices. Selecting the right nanoparticle type involves balancing synthesis techniques and biocompatibility to optimize therapeutic outcomes. Properly synthesized and biocompatibility-tested nanoparticles increase the efficacy and safety of PTT, making them indispensable for successful treatment. Additionally, ongoing research into AI security enhances the development and testing of safer nanomaterials for medical applications.

Magnetic Nanoparticle-Based Hybrid Materials: Fundamentals and Applications (Woodhead Publishing Series in Electronic and Optical Materials)
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
Mechanisms of Light-to-Heat Conversion

The effectiveness of nanoparticle-mediated photothermal therapy hinges on how efficiently nanoparticles convert incident light into heat. This process depends on their photothermal efficiency, which determines how much light absorption results in heat generation. When nanoparticles absorb light, their electrons are excited to higher energy states. As these electrons relax back, energy is released as heat, raising the local temperature. Factors influencing this include nanoparticle composition, size, and shape, which affect their light absorption properties. Enhancing photothermal efficiency can significantly improve therapeutic outcomes by maximizing heat generation from absorbed light.

Nanoparticle-Mediated Targeted Drug Delivery Systems
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
Targeting Strategies for Tumor Specificity
To improve tumor specificity in photothermal therapy, you can use active targeting ligands that bind directly to tumor markers. Additionally, stimuli-responsive activation allows nanoparticles to become active only in the tumor environment, reducing off-target effects. Combining these strategies enhances treatment precision and minimizes damage to healthy tissue. Incorporating best practices from successful targeting techniques further optimizes therapeutic outcomes.
Active Targeting Ligands
Active targeting ligands play a crucial role in enhancing the specificity of nanoparticle-mediated photothermal therapy by directing nanoparticles directly to tumor cells. You achieve this through ligand conjugation, which attaches targeting molecules to nanoparticle surfaces, ensuring they bind selectively. Receptor specificity is key; ligands are chosen to recognize overexpressed receptors on tumor cells, minimizing damage to healthy tissue. This targeted approach improves therapeutic efficiency and reduces side effects. When designing ligands, consider their binding affinity, stability, and biocompatibility. Successful ligand conjugation ensures precise delivery, allowing for effective photothermal conversion at the tumor site. By focusing on receptor specificity, you can optimize nanoparticle performance for personalized, highly selective cancer treatments.
- Ligand conjugation techniques
- Receptor overexpression targeting
- Enhanced tumor cell recognition
- Improved therapeutic precision
Stimuli-Responsive Activation
Stimuli-responsive activation enhances nanoparticle-mediated photothermal therapy by guaranteeing nanoparticles become active only in the presence of specific tumor microenvironment cues. You leverage temperature sensitivity so that nanoparticles activate when tumor tissue reaches a certain temperature during heating, minimizing damage to healthy tissue. Chemical triggers, such as pH changes or enzyme presence, can also initiate nanoparticle activation selectively within tumors. This targeted approach prevents premature activation elsewhere in the body, increasing treatment safety and efficacy. By designing nanoparticles responsive to these stimuli, you improve tumor specificity and control over therapy delivery. Ultimately, stimuli-responsive activation ensures that the photothermal effect occurs only under desired conditions, optimizing therapeutic outcomes and reducing side effects.

Simple HealthKit At-Home Common STD Test Kit for Chlamydia, Gonorrhea & Trichomoniasis – Tests for the Most Common STDs – Free Follow-Up/Telehealth & High Quality Lab Results
Tests for the Most Common STDs: An easy-to-use common STD test with simple, high quality accurate, and private…
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
Advantages Over Conventional Therapies
Nanoparticle-mediated photothermal therapy offers you enhanced targeting precision, ensuring the treatment focuses directly on tumor cells. This accuracy helps minimize side effects often seen with traditional therapies. As a result, you get a more effective treatment with fewer risks to healthy tissue. Additionally, the technology’s ability to leverage private placement strategies can facilitate funding for innovative medical research and development.
Enhanced Targeting Precision
Unlike traditional therapies, nanoparticle-mediated photothermal therapy offers substantially enhanced targeting precision by allowing you to direct treatment specifically to cancer cells. This precision relies on nanoparticles’ ability to perform efficient photothermal conversion, converting light into heat precisely where needed. You can also leverage immune modulation to stimulate your immune system, boosting the body’s natural defenses against cancer. This targeted approach reduces damage to healthy tissue and improves treatment outcomes. The nanoparticles can be engineered to recognize specific cancer markers, ensuring accurate delivery. Additionally, their ability to focus heat on tumor sites minimizes collateral effects. Overall, this method provides a more precise, effective, and adaptable cancer treatment, aligning therapy directly with your tumor’s unique characteristics.
- Specific recognition of cancer markers
- Precise heat delivery through photothermal conversion
- Immune modulation enhances immune response
- Reduced damage to healthy tissue
Minimized Side Effects
Because nanoparticle-mediated photothermal therapy targets cancer cells precisely, it considerably reduces the risk of damaging healthy tissue compared to conventional treatments. This focused approach minimizes nanoparticle toxicity, lowering the chance of adverse effects often seen with traditional therapies. Additionally, since the therapy activates only at the tumor site, it triggers a limited immune response, reducing systemic side effects like inflammation. Unlike chemotherapy or radiation, which can affect multiple organs, this method spares healthy cells, leading to fewer complications. The controlled delivery and localized heating also help prevent collateral tissue damage. Overall, this targeted strategy offers a safer alternative, providing effective cancer treatment with minimized side effects and improved patient comfort.
Challenges and Limitations in PTT
While nanoparticle-mediated photothermal therapy offers promising treatment avenues, several challenges hinder its widespread clinical adoption. You may encounter biocompatibility concerns, as not all nanoparticles are fully safe or tolerated in the body. Manufacturing challenges also pose obstacles, including ensuring consistent quality and scalability. Additionally, controlling nanoparticle distribution and accumulation at tumor sites remains difficult, affecting treatment efficacy. Safety and regulatory hurdles further slow progress, requiring extensive testing to confirm long-term safety. Finally, optimizing nanoparticle properties for targeting and stability without compromising biocompatibility adds complexity. Overcoming these issues is essential for translating PTT from promising research to reliable clinical practice. Addressing these challenges will pave the way for safer, more effective photothermal therapies. Advanced nanoparticle design can help improve targeting and reduce side effects.
Recent Advances and Future Directions

Recent advances in nanoparticle-mediated photothermal therapy (PTT) have markedly improved its potential as a viable cancer treatment. Researchers are developing new nanoparticles with enhanced targeting abilities and better biocompatibility, addressing concerns about nanoparticle toxicity. These innovations aim to maximize therapeutic efficacy while minimizing side effects. However, regulatory hurdles remain a significant challenge; gaining approval requires thorough safety evaluations and standardized manufacturing processes. Progress is being made in understanding long-term toxicity and environmental impact, which will help streamline approval pathways. Future directions focus on designing smarter, more biocompatible nanoparticles, along with establishing clear guidelines for clinical translation. These efforts will help overcome current limitations, bringing nanoparticle-mediated PTT closer to widespread clinical application and improved patient outcomes.
Clinical Applications and Case Studies
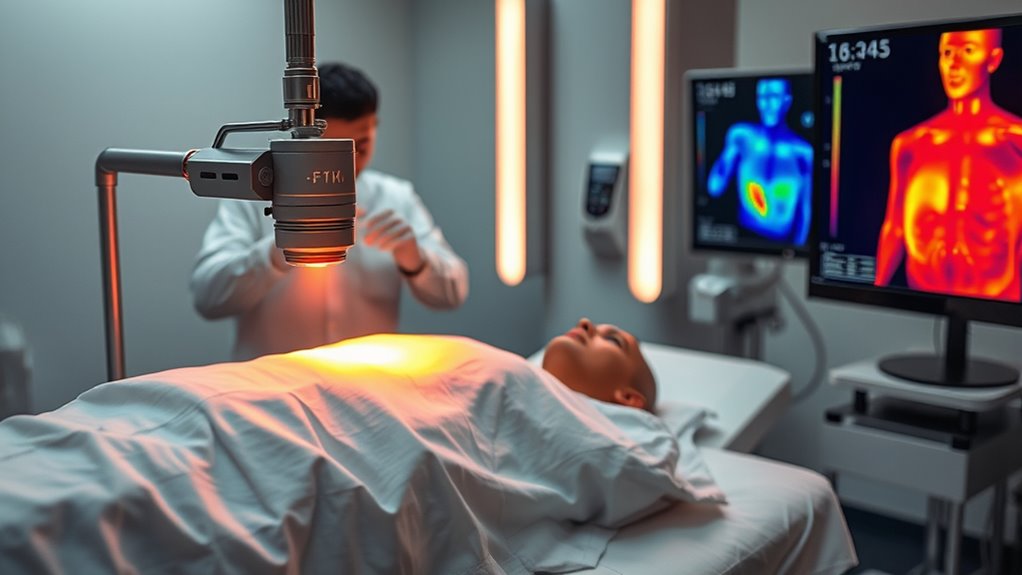
Advances in nanoparticle design have paved the way for their application in clinical settings, transforming promising laboratory results into real-world cancer treatments. You’ll find that clinical case studies highlight improved patient outcomes, showcasing the potential of nanoparticle-mediated photothermal therapy. However, regulatory hurdles remain a challenge, slowing widespread adoption. Additionally, understanding small mistakes, big impact in the development process can help mitigate risks and improve treatment safety. Key aspects include:
Nanoparticle advances are transforming lab results into real-world cancer treatments, but regulatory hurdles slow adoption.
- Successes in targeting difficult tumors with minimal side effects
- Variability in patient responses, emphasizing personalized approaches
- Ongoing trials demonstrating safety and efficacy
- Regulatory approval processes influencing clinical implementation
While these therapies show promise, steering regulatory pathways is vital to ensure safety and efficacy. Your understanding of these applications can help in advancing nanoparticle-based treatments from research to routine clinical practice.
Frequently Asked Questions
How Do Nanoparticles Affect Surrounding Healthy Tissues During PTT?
During photothermal therapy, you might worry about off target effects on healthy tissues. Nanoparticles are designed for tissue specificity, which helps target cancer cells precisely. However, if the nanoparticles aren’t perfectly selective, they can accumulate in healthy tissues, causing unintended heating and damage. To minimize these risks, researchers focus on improving targeting accuracy, ensuring the therapy remains effective on cancer while sparing surrounding healthy tissues.
What Are the Long-Term Biocompatibility Concerns of Nanoparticle Use?
You should consider that long-term biocompatibility concerns include systemic toxicity and long-term accumulation of nanoparticles in your body. These particles might persist in tissues, potentially causing inflammation or damage over time. It is crucial to evaluate how your body clears these materials and whether they build up, which could lead to adverse effects. Ongoing research aims to improve nanoparticle design for safer, more biocompatible treatments in the future.
How Is PTT Integrated With Other Cancer Treatments?
Think of PTT as a powerful team player, working alongside other cancer treatments. You combine strategies like chemotherapy, radiotherapy, or immunotherapy to create a synergistic effect, enhancing overall effectiveness. This integration allows PTT to target tumors more precisely, reducing side effects and increasing success rates. By leveraging combination strategies, you optimize treatment outcomes, making PTT a versatile and valuable component in extensive cancer therapy plans.
Are There Any Known Resistance Mechanisms to Nanoparticle-Mediated PTT?
Yes, resistance mechanisms to nanoparticle-mediated PTT exist. Your tumor microenvironment can limit therapy effectiveness through hypoxia or high antioxidant levels, reducing heat-induced cell death. Additionally, tumor cells may alter their surface properties, hindering nanoparticle uptake. To overcome this, you can modify nanoparticles to improve targeting or penetration, and consider strategies that modify the tumor microenvironment, enhancing overall PTT efficacy and reducing resistance development.
What Are the Regulatory Hurdles for Clinical Approval of Nanoparticle-Based PTT?
Think of regulatory hurdles as towering gates you must pass through to bring your nanoparticle-based PTT to patients. You’ll face complex regulatory pathways involving safety, efficacy, and manufacturing standards, creating approval challenges. These hurdles require thorough data and rigorous testing to satisfy agencies like the FDA or EMA. Piloting this maze demands patience, meticulous documentation, and strategic planning, but overcoming it means transforming innovative therapy into a trusted clinical reality.
Conclusion
As you explore nanoparticle-mediated photothermal therapy, remember it’s a beacon of hope guiding cancer’s darkness. The light you harness transforms shadows into heat, targeting tumors with precision. Though challenges remain like clouds before the sun, ongoing advances promise a brighter future. With each breakthrough, you’re part of a journey—turning tiny particles into powerful warriors. Together, you and this technology illuminate a path toward hope, healing, and a new dawn in cancer treatment.