Nanobots are tiny machines made of biocompatible materials like DNA, proteins, or polymers. They move inside your body using external magnetic fields, ultrasound, or chemical reactions that generate propulsion. They find their targets by sensing environmental cues or attaching to specific cells. Once in place, they deliver medicine or perform repairs. Curious about how researchers are overcoming challenges? Keep exploring to discover the future of nanobots.
Key Takeaways
- Nanobots are tiny devices made from biocompatible materials that can perform tasks inside the body.
- They move using external magnetic fields, ultrasound waves, or chemical reactions to navigate tissues.
- Equipped with sensors and tools, they target specific cells or areas for diagnosis or drug delivery.
- Power is supplied externally through magnetic, ultrasound, or light signals, controlling their movement and actions.
- They work by sensing environmental cues, following signals, and responding with precise movements to achieve medical goals.
nanobot drug delivery systems
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
What Are Nanobots Made Of?

Nanobots are built from a variety of biocompatible and synthetic materials that enable their functions inside the body. You’ll find they often include DNA, proteins, or synthetic polymers to guarantee they’re safe and compatible with biological systems. Their structures can feature tiny components like sensors, actuators, drug reservoirs, and onboard processors, all miniaturized to fit within a few micrometers. For example, some use flexible gold-silver-nickel nanowires or Janus capsules with a silica core. Many nanobots have a helical shape combined with magnetic materials, allowing them to move when exposed to external magnetic fields. These materials work together to make nanobots versatile, durable, and capable of performing complex tasks, from drug delivery to precise targeting within the body. Material composition plays a crucial role in determining their functionality and safety. Additionally, advances in biocompatibility ensure these nanobots can operate safely over extended periods without adverse immune responses. Furthermore, ongoing research into nanoengineering enhances their ability to perform increasingly sophisticated operations within biological environments. The tiny components used in their construction are carefully designed to maximize efficiency and minimize potential risks. Developing nanomaterial stability is also essential to ensure they remain functional under various conditions inside the body.
biocompatible nanobots for medical use
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
How Do Nanobots Move Inside the Body?
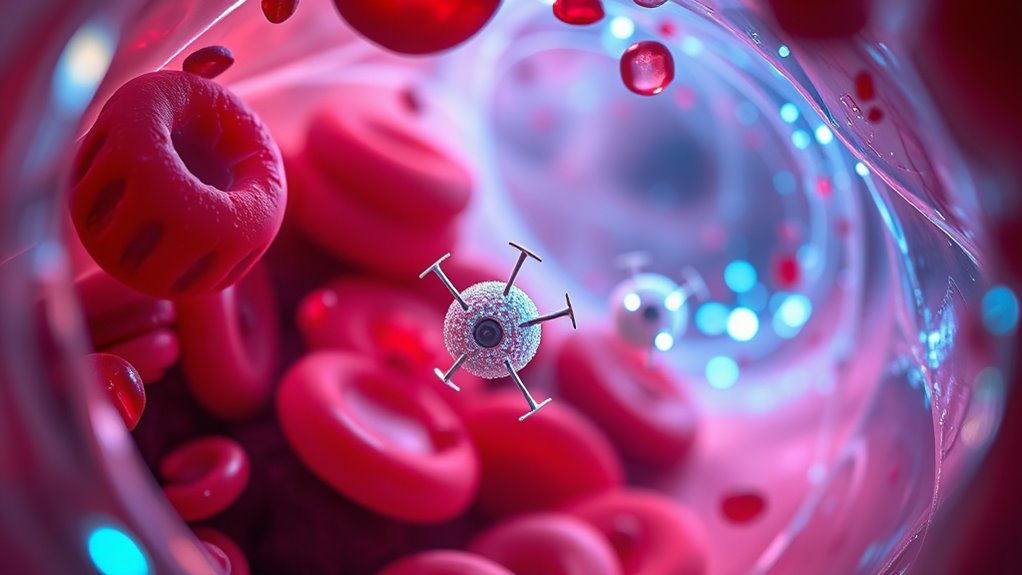
Have you ever wondered how tiny robots navigate through the complex environment of the human body? Nanobots move using various methods designed for their small size and environment. Many use external magnetic fields to rotate their helical bodies, propelling them forward much like a propeller. Others harness chemical reactions, such as breaking down urea or hydrogen peroxide, creating fluid flows or gas bubbles that push them along. Ultrasound waves can also direct nanobots by generating pressure gradients, helping them penetrate tissues or reach specific sites. Some are inspired by natural structures like flagella or cilia, vibrating or waving to move through fluids. These movement strategies allow nanobots to travel precisely inside the body, targeting areas for drug delivery, diagnostics, or treatment. At‑home beauty tech devices also explore some of these principles to improve skincare delivery systems. Additionally, understanding the energetic alignment of nanobots with their environment can optimize their efficiency and effectiveness in medical applications, especially when considering smart home integration for remote control and monitoring. Exploring biocompatible materials is essential to ensure nanobots operate safely within the human body without causing adverse reactions, and advances in material science are significantly contributing to these developments.
nanobot medical repair devices
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
What Powers Nanobots’ Movement?

The power behind nanobots’ movement comes from various energy sources and mechanisms that activate their miniature components. You can think of it like different fuels or signals that make them move. Some nanobots use magnetic fields to spin their helical bodies, pushing them forward. Others rely on chemical reactions, such as enzymes breaking down substances to generate movement. External stimuli like ultrasound or light can also trigger motion, causing nanobots to shift direction or speed. Here’s a quick look at their power sources:
| Power Source | How It Works |
|---|---|
| Magnetic Fields | Spin or rotate nanobots via external magnets |
| Chemical Reactions | Catalyze substances to produce propulsion |
| Ultrasound | Use sound waves to push or steer nanobots |
| Light (UV, IR) | Trigger conformational changes for movement |
| External Electric Fields | Generate motion through electrical stimulation |
These mechanisms make nanobots versatile and precise in their motion. Many of these energy sources are harnessed from external environmental signals, which enable nanobots to operate efficiently in various settings. Additionally, advances in energy conversion techniques are expanding the capabilities of nanobots, allowing for more complex and controlled movements.
magnetic nanobot navigation tools
As an affiliate, we earn on qualifying purchases.
As an affiliate, we earn on qualifying purchases.
How Do Nanobots Find Their Targets?

Nanobots find their targets by using specialized recognition strategies that identify specific cells or chemicals. They navigate through complex environments with guidance systems like magnetic fields or chemical cues. Understanding these methods reveals how nanobots reach precise locations to perform their tasks efficiently. Additionally, innovations in nanotechnology continue to enhance their targeting accuracy and operational capabilities. Proper calibration and diagnostics are also crucial to ensure nanobots operate correctly within their designated environments. Advances in biological recognition improve their ability to distinguish between healthy and diseased cells, further increasing their effectiveness, especially as researchers develop more targeted approaches to improve treatment precision. Incorporating sensor technology allows nanobots to adapt in real-time, thereby increasing their success rate in complex biological settings.
Target Recognition Strategies
Target recognition strategies enable nanobots to locate specific cells or tissues within the body with high precision. You can program nanobots to identify targets by attaching specialized molecules like antibodies, aptamers, or DNA sequences that bind only to specific cell markers. These molecules act as biological “locks,” ensuring the nanobots stick to their intended targets and avoid healthy tissue. Some nanobots also sense chemical signals or changes in the environment, such as pH or enzyme presence, to detect diseased areas. By combining these recognition techniques, nanobots can accurately distinguish cancer cells, inflamed tissue, or other specific sites. This targeted approach maximizes effectiveness while minimizing side effects, making nanobots highly efficient tools for precise diagnosis and therapy. Additionally, advances in target recognition improve the ability of nanobots to differentiate between healthy and abnormal cells more reliably. Furthermore, ongoing innovations in biomolecular attachment enhance the stability and specificity of these recognition molecules, leading to even more precise targeting. These improvements also contribute to reducing false positives, ensuring that nanobots respond only to true disease markers. The integration of multiple recognition methods allows for a combination of techniques, improving overall accuracy and robustness of nanobot targeting. As research progresses, the integration of multiple recognition methods allows for even greater targeting accuracy and reduces the likelihood of false positives.
Navigation and Guidance
How do nanobots navigate complex biological environments to reach their specific targets? They use a combination of external guidance and programmed responses. Magnetic nanobots, for example, rotate in magnetic fields to swim precisely through blood or tissue. Ultrasound waves can steer nanobots by creating pressure gradients, helping them penetrate cell layers. Some nanobots are designed to follow chemical signals, like enzymes or pH changes, guiding them toward inflamed or cancerous tissues. Others hitch a ride on migrating cells or attach to specific receptors, using biological cues for direction. Researchers also program nanobots with DNA or molecular markers that respond to environmental signals, ensuring they find their targets accurately. This mix of external control and biological sensing makes navigation possible even in complex, crowded environments. Additionally, advances in targeted delivery techniques continue to improve the precision of nanobot navigation in medical applications. New methods involving environmental sensing allow nanobots to adapt dynamically to their surroundings, enhancing their ability to reach precise locations, and ongoing research explores autonomous decision-making to further improve navigation accuracy.
How Do Nanobots Deliver Medicine?

Ever wondered how tiny robots can deliver medicine precisely where it’s needed? Nanobots navigate through your body to target specific sites, releasing drugs exactly when and where they should. They use various mechanisms for delivery:
- Attach to or enter cells with specialized sensors.
- Carry drug reservoirs that release medication upon reaching the target.
- Use magnetic or ultrasound signals to trigger drug release.
- Follow environmental cues like chemical signals or pH changes.
- Swarm together for collective delivery in complex tissues.
These features guarantee that medicine isn’t dispersed everywhere but focused on the problem area, reducing side effects and increasing effectiveness. By combining navigation, sensing, and controlled release, nanobots revolutionize targeted therapy, making treatments more precise and personalized.
What Are Some Uses of Nanobots in Medicine?

Nanobots are transforming medicine by performing a variety of innovative tasks inside the body. You might use them to target specific areas, like delivering drugs directly to tumors or repairing tissues. They can detect infections or chemical imbalances, providing quick diagnostics. Some nanobots carry medication, releasing it precisely where needed, reducing side effects. Others help remove blood clots or clear arterial blockages, improving blood flow. Here’s a quick look at their uses:
| Application | Function |
|---|---|
| Targeted drug delivery | Delivering medication directly to disease sites |
| Diagnostic sensing | Detecting infections or chemical imbalances |
| Blood clot removal | Clearing blockages to restore circulation |
These tiny tools are revolutionizing healthcare by enhancing precision and effectiveness.
How Are Nanobots Controlled and Directed?

Controlling and directing nanobots relies on external energy sources that influence their movement and behavior. You can guide them with magnetic fields, ultrasound, or light, depending on their design. Magnetic fields rotate helical structures, creating torque that propels the nanobots forward. Ultrasound generates pressure gradients, pushing or pulling nanobots through tissues. Light triggers conformational changes in photo-responsive materials, enabling precise control. External chemical reactions, like enzyme activity, can also power motion. Additionally, nanobots can be programmed to respond to specific environmental cues or attach to target cells for targeted delivery. Here’s how control is achieved:
External energy sources like magnetic fields, ultrasound, and light enable precise nanobot control and targeted delivery.
- Magnetic fields manipulate magnetic or ferromagnetic components.
- Ultrasound induces directional pressure for movement.
- Light causes shape changes in photo-sensitive parts.
- Chemical stimuli trigger catalysis-driven propulsion.
- Environmental cues activate specific behaviors.
What Are Examples of Current Nanobot Projects?

Current nanobot projects showcase a variety of innovative designs and functionalities that demonstrate the technology’s potential. For example, xenobots are tiny, living robots made from about 500-1000 cells, capable of moving in straight lines or circles for up to 10 days, showing promise for medical applications. Helical magnetic nanomotors are used to manipulate cells inside the body, providing targeted treatment options. Other projects include nanoswimmers with undulating, S-shaped motions controlled by magnetic fields, mimicking biological movement. Fish-inspired nanorobots with gold heads and nickel bodies aim to navigate complex biological environments. Researchers use optical microscopy and PET imaging to visualize these movements in real-time, advancing our understanding of nanobot capabilities and paving the way for future medical breakthroughs.
What Challenges Do Researchers Face With Nanobots?

You’ll find that ensuring nanobots are biocompatible is a major hurdle, as they must avoid harming human tissues while still functioning effectively. Maneuvering precisely through complex biological environments proves tricky, especially when controlling their movement in real time. Plus, powering these tiny machines remains a challenge, since they need reliable energy sources without adding bulk or toxicity.
Biocompatibility Issues
Researchers face significant challenges in ensuring nanobots are biocompatible, as their tiny size and complex components can trigger immune responses or toxicity. Your body might see these devices as threats, leading to inflammation or rejection. Achieving safe interactions involves careful material selection and surface modifications. You also need to prevent harmful reactions from degradation products or unintended interactions with cells. The design must balance functionality with safety, often requiring extensive testing.
- Using materials like DNA, proteins, or synthetic polymers minimizes immune reactions
- Coating nanobots with biocompatible substances reduces toxicity
- Avoiding non-degradable or toxic metals prevents harmful buildup
- Designing for controlled degradation ensures safe elimination
- Balancing functionality with minimal immune activation remains a key challenge
Precise Navigation Difficulties
Achieving precise navigation of nanobots within complex biological environments remains a significant challenge due to their tiny size and the dynamic nature of bodily tissues. You must contend with unpredictable fluid flows, tissue heterogeneity, and immune responses that can divert or destroy nanobots. External control methods like magnetic fields or ultrasound help, but their accuracy diminishes in dense or complex areas. To illustrate, consider the following:
| Challenge | Effect on Navigation |
|---|---|
| Tissue heterogeneity | Causes unpredictable movement |
| Fluid flow variability | Alters nanobot paths |
| Immune system interference | Risks nanobot clearance or damage |
| Limited sensor precision | Reduces targeting accuracy |
| External control limitations | Decreases maneuverability |
These factors make it difficult to guide nanobots reliably to specific targets.
Power Supply Limitations
Power supply limitations pose a significant hurdle for nanobot functionality, especially given their tiny size and complex environments. You can’t easily include large batteries or power sources inside such small devices. Researchers face challenges like:
- Miniaturizing energy storage to fit within nanometer scales
- Ensuring consistent power delivery in dynamic biological environments
- Developing sustainable, biocompatible energy sources
- Overcoming energy losses during transmission or conversion
- Maintaining power without generating harmful heat or side effects
These issues make it difficult to enable nanobots to operate continuously or perform complex tasks. Finding lightweight, efficient, and safe power solutions remains a vital obstacle. Overcoming these limitations is essential for advancing nanobot applications in medicine and other fields.
Frequently Asked Questions
Can Nanobots Operate Autonomously Without Human Control?
Yes, nanobots can operate autonomously without human control. They use onboard sensors and processing units to detect environmental signals, allowing them to navigate, target specific cells, or deliver drugs independently. External energy sources like magnetic fields, ultrasound, or chemical reactions also help control their movement. By responding to these stimuli, nanobots perform tasks precisely, making them effective for medical applications without needing direct human intervention at every step.
How Long Can Nanobots Function Inside the Human Body?
You can expect nanobots to function inside your body for hours to days, depending on their design and purpose. They often have biocompatible materials that prevent rejection, and their energy sources, like magnetic or chemical triggers, influence their lifespan. Some are engineered for short-term tasks, while others are designed for longer deployments, but factors like immune responses and material durability ultimately determine how long they stay active.
Are Nanobots Safe or Do They Pose Health Risks?
Nanobots can be safe when properly designed and tested, but they might pose health risks if they cause immune reactions or accumulate in the body. You should guarantee they use biocompatible materials like DNA or proteins and undergo rigorous testing. With controlled navigation via magnetic fields or ultrasound, you can minimize dangers. Overall, safety depends on careful engineering, thorough research, and monitoring during use.
How Are Nanobots Powered or Recharged During Operation?
You power nanobots using external energy sources like magnetic fields, ultrasound, or light, which activate their movement and functions. As they operate, they don’t require traditional recharging; instead, these energy inputs continuously drive their propulsion and actuation. You can control their navigation precisely by adjusting these external stimuli, ensuring they stay active and effective during tasks like targeted drug delivery or diagnostics without needing to recharge like batteries.
Will Nanobots Be Affordable for Widespread Medical Use?
Nanobots are becoming more affordable for widespread medical use as technology advances and production costs decrease. You’ll see them integrated into healthcare because of innovations in biocompatible materials and scalable manufacturing processes. As research progresses, expect prices to drop further, making nanobots accessible for routine treatments, targeted drug delivery, and diagnostics. This affordability will enable you to benefit from personalized, minimally invasive therapies, revolutionizing healthcare affordability and accessibility worldwide.
Conclusion
Imagine a future where nanobots could deliver medicine precisely to cancer cells, reducing side effects. Currently, scientists are developing over 1,000 nanobot projects worldwide, aiming to revolutionize healthcare. As you learn how these tiny machines work, you realize their potential to save lives and improve treatments. While challenges remain, ongoing research continues to push boundaries. With such rapid progress, the era of nanobot-powered medicine might be closer than you think.